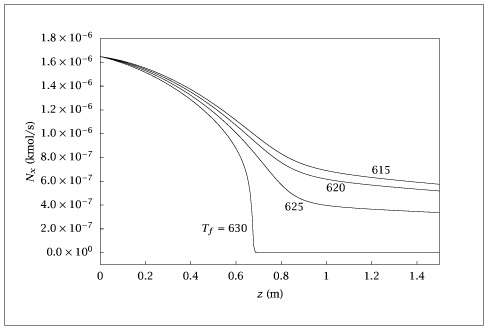
Figure 6.35:
Molar flow of o-xylene versus reactor length for different feed temperatures.
Code for Figure 6.35
Text of the GNU GPL.
main.py
1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
53
54 | # Converted from xyleneT.m - o-xylene oxidation PFR temperature profiles
import numpy as np
from scipy.integrate import solve_ivp
from misc import save_ascii
R_tube = 1.25e-2 # tube radius, m
Ac = np.pi*R_tube**2
Qrho = 0.0026371 # mass flow, kg/sec
Pf = 1.01e2 # feed pressure, kN/m^2
Mwf = 0.98*(0.79*28+0.21*32)+0.02*106.17 # mol wt feed, kg/kmol
Tf = 625. # feed temperature, K
Rg = 8.314 # kJ/(K kmol)
Nf = Qrho / Mwf # kmol/sec
E = 13636. # K
Tm = 625. # K
km = 2.0822 # 1/sec
Ta = 625. # K
Cp = 0.992 # kJ/kg K
delH = -1.284e6 # kJ/kmol
U = 0.373 # kJ/(m^2 sec K)
beta = delH * Ac / (Qrho*Cp)
Gamma = 2*np.pi*R_tube*U / (Qrho*Cp)
l = 1.5 # m
p = dict(Nf=Nf, Pf=Pf, Rg=Rg, E=E, Tm=Tm, km=km, Ta=Ta, beta=beta, Gamma=Gamma, Ac=Ac)
def pfr(t, x, p):
Nx = x[0]; T = x[1]
Q = p['Nf'] / (p['Pf']/(p['Rg']*T))
cx = Nx / Q
k = p['km'] * np.exp(-p['E']*(1./T - 1./p['Tm']))
rate = k * cx
return [-rate*p['Ac'],
-p['beta']*rate + p['Gamma']*(p['Ta']-T)]
npts = 200
z = np.linspace(0., l, npts)
yxfeed = 0.019
Nxf = yxfeed * Nf
Tfeed = [615., 620., 625., 630.]
nfeed = len(Tfeed)
yx_all = np.zeros((npts, nfeed))
T_all = np.zeros((npts, nfeed))
eps_sq = np.sqrt(np.finfo(float).eps)
for i, Tf_i in enumerate(Tfeed):
x0_i = [Nxf, Tf_i]
sol = solve_ivp(lambda t, x: pfr(t, x, p), [0., l], x0_i,
method='Radau', t_eval=z, rtol=eps_sq, atol=eps_sq)
yx_all[:, i] = sol.y[0, :]
T_all[:, i] = sol.y[1, :]
table = np.column_stack([z, yx_all, T_all])
save_ascii('xyleneT.dat', table)
|
